Description







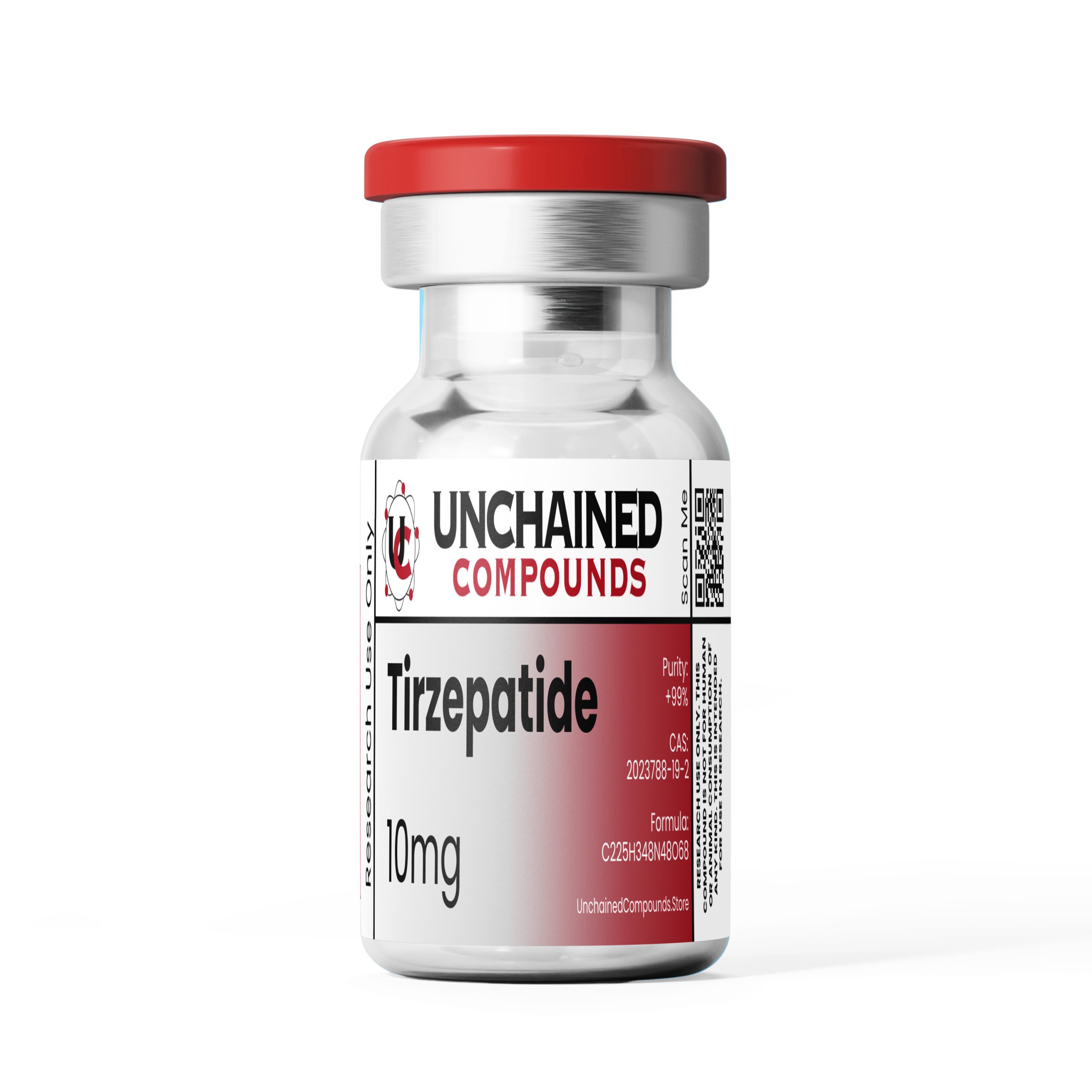
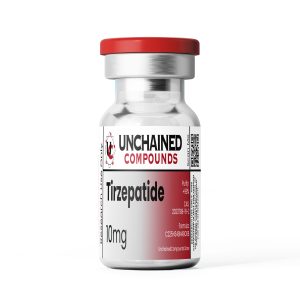
Tirzepatide is a synthetic derivative of gastric inhibitory polypeptide (GIP) that has simultaneous glucagon-like peptide-1 (GLP-1) functionality as well. This combination allows Tirzepatide to lower blood glucose levels, increase insulin sensitivity, boost feelings of satiety, and accelerate weight loss.
Specifications
- Chemical Formula: C225H348N48O68
- Molecular Mass: 4813g/mol
- Synonyms: Tirzepatide (LY3298176) 2023788-19-2 Tirzepatide GTPL11429
- CAS Number: 2023788-19-2
- PubChem: 156588324
- Total Amount of the Active Ingredient: 10 mg (1 vial)
- Shelf Life: 36 months
Product Quality
Lab tests are occasionally published on the website. You can have the product you bought from us tested at any HPLC licensed testing facility and if the results are negative, we will refund the following:
- Cost of HPLC test
- Total amount of the order + shipping fee
Research Studies
Tirzepatide Once Weekly for the Treatment of Obesity
Abstract
BACKGROUND
Obesity is a chronic disease that results in substantial global morbidity and mortality. The efficacy and safety of tirzepatide, a novel glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 receptor agonist, in people with obesity are not known.
METHODS
In this phase 3 double-blind, randomized, controlled trial, we assigned 2539 adults with a body-mass index (BMI; the weight in kilograms divided by the square of the height in meters) of 30 or more, or 27 or more and at least one weight-related complication, excluding diabetes, in a 1:1:1:1 ratio to receive once-weekly, subcutaneous tirzepatide (5 mg, 10 mg, or 15 mg) or placebo for 72 weeks, including a 20-week dose-escalation period. Coprimary end points were the percentage change in weight from baseline and a weight reduction of 5% or more. The treatment-regimen estimand assessed effects regardless of treatment discontinuation in the intention-to-treat population.
RESULTS
At baseline, the mean body weight was 104.8 kg, the mean BMI was 38.0, and 94.5% of participants had a BMI of 30 or higher. The mean percentage change in weight at week 72 was −15.0% (95% confidence interval [CI], −15.9 to −14.2) with 5-mg weekly doses of tirzepatide, −19.5% (95% CI, −20.4 to −18.5) with 10-mg doses, and −20.9% (95% CI, −21.8 to −19.9) with 15-mg doses and −3.1% (95% CI, −4.3 to −1.9) with placebo (P<0.001 for all comparisons with placebo). The percentage of participants who had weight reduction of 5% or more was 85% (95% CI, 82 to 89), 89% (95% CI, 86 to 92), and 91% (95% CI, 88 to 94) with 5 mg, 10 mg, and 15 mg of tirzepatide, respectively, and 35% (95% CI, 30 to 39) with placebo; 50% (95% CI, 46 to 54) and 57% (95% CI, 53 to 61) of participants in the 10-mg and 15-mg groups had a reduction in body weight of 20% or more, as compared with 3% (95% CI, 1 to 5) in the placebo group (P<0.001 for all comparisons with placebo). Improvements in all prespecified cardiometabolic measures were observed with tirzepatide. The most common adverse events with tirzepatide were gastrointestinal, and most were mild to moderate in severity, occurring primarily during dose escalation. Adverse events caused treatment discontinuation in 4.3%, 7.1%, 6.2%, and 2.6% of participants receiving 5-mg, 10-mg, and 15-mg tirzepatide doses and placebo, respectively.
CONCLUSIONS
In this 72-week trial in participants with obesity, 5 mg, 10 mg, or 15 mg of tirzepatide once weekly provided substantial and sustained reductions in body weight. (Supported by Eli Lilly; SURMOUNT-1 ClinicalTrials.gov number, NCT04184622.)
Tirzepatide: A Systematic Update
Abstract
Tirzepatide is a new molecule capable of controlling glucose blood levels by combining the dual agonism of Glucose-Dependent Insulinotropic Polypeptide (GIP) and Glucagon-Like Peptide-1 (GLP-1) receptors. GIP and GLP1 are incretin hormones: they are released in the intestine in response to nutrient intake and stimulate pancreatic beta cell activity secreting insulin. GIP and GLP1 also have other metabolic functions. GLP1, in particular, reduces food intake and delays gastric emptying. Moreover, Tirzepatide has been shown to improve blood pressure and to reduce Low-Density Lipoprotein (LDL) cholesterol and triglycerides. Tirzepatide efficacy and safety were assessed in a phase III SURPASS 1-5 clinical trial program. Recently, the Food and Drug Administration approved Tirzepatide subcutaneous injections as monotherapy or combination therapy, with diet and physical exercise, to achieve better glycemic blood levels in patients with diabetes. Other clinical trials are currently underway to evaluate its use in other diseases. The scientific interest toward this novel, first-in-class medication is rapidly increasing. In this comprehensive and systematic review, we summarize the main results of the clinical trials investigating Tirzepatide and the currently available meta-analyses, emphasizing novel insights into its adoption in clinical practice for diabetes and its future potential applications in cardiovascular medicine.
Abstract
Background: Weight reduction is essential for improving health outcomes in people with obesity and type 2 diabetes. We assessed the efficacy and safety of tirzepatide, a glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 receptor agonist, versus placebo, for weight management in people living with obesity and type 2 diabetes.
Methods: This phase 3, double-blind, randomised, placebo-controlled trial was conducted in seven countries. Adults (aged ≥18 years) with a body-mass index (BMI) of 27 kg/m2 or higher and glycated haemoglobin (HbA1c) of 7-10% (53-86 mmol/mol) were randomly assigned (1:1:1), using a computer-generated random sequence via a validated interactive web-response system, to receive either once-weekly, subcutaneous tirzepatide (10 mg or 15 mg) or placebo for 72 weeks. All participants, investigators, and the sponsor were masked to treatment assignment. Coprimary endpoints were the percent change in bodyweight from baseline and bodyweight reduction of 5% or higher. The treatment-regimen estimand assessed effects regardless of treatment discontinuation or initiation of antihyperglycaemic rescue therapy. Efficacy and safety endpoints were analysed with data from all randomly assigned participants (intention-to-treat population). This trial is registered with ClinicalTrials.gov, NCT04657003.
Findings: Between March 29, 2021, and April 10, 2023, of 1514 adults assessed for eligibility, 938 (mean age 54·2 years [SD 10·6], 476 [51%] were female, 710 [76%] were White, and 561 [60%] were Hispanic or Latino) were randomly assigned and received at least one dose of tirzepatide 10 mg (n=312), tirzepatide 15 mg (n=311), or placebo (n=315). Baseline mean bodyweight was 100·7 kg (SD 21·1), BMI 36·1 kg/m2 (SD 6·6), and HbA1c 8·02% (SD 0·89; 64·1 mmol/mol [SD 9·7]). Least-squares mean change in bodyweight at week 72 with tirzepatide 10 mg and 15 mg was -12·8% (SE 0·6) and -14·7% (0·5), respectively, and -3·2% (0·5) with placebo, resulting in estimated treatment differences versus placebo of -9·6% percentage points (95% CI -11·1 to -8·1) with tirzepatide 10 mg and -11·6% percentage points (-13·0 to -10·1) with tirzepatide 15 mg (all p<0·0001). More participants treated with tirzepatide versus placebo met bodyweight reduction thresholds of 5% or higher (79-83% vs 32%). The most frequent adverse events with tirzepatide were gastrointestinal-related, including nausea, diarrhoea, and vomiting and were mostly mild to moderate in severity, with few events leading to treatment discontinuation (<5%). Serious adverse events were reported by 68 (7%) participants overall and two deaths occurred in the tirzepatide 10 mg group, but deaths were not considered to be related to the study treatment by the investigator.
Interpretation: In this 72-week trial in adults living with obesity and type 2 diabetes, once-weekly tirzepatide 10 mg and 15 mg provided substantial and clinically meaningful reduction in bodyweight, with a safety profile that was similar to other incretin-based therapies for weight management.
Shipping
USA
Canada
If your shipment was seized (International Orders), we will provide a 50% discount applicable on your next purchase. Please contact us for more information.
Disclaimer
The information provided above is not intended to substitute medical advice, diagnosis, or treatment. Should you have any questions regarding a medical condition, seek the advice of your physician or a qualified healthcare provider. In no case should medical advice be disregarded or delayed because of what you have read or seen. We bear no responsibility or liability for your use of any of our research compounds and products. Please note that they are being sold for research purposes ONLY. We do NOT condone any personal use.
NOTE: In some cases wherein the assigned top colors are out of stock, a different top color will be used to ensure that your order will not be delayed. Should you need assistance identifying the peptide vial that you received, please send us an email at support@unchainedcompounds.store.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are intended for in-vitro studies only. In-vitro studies (Latin: “in glass”) are performed outside the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat, or cure any medical condition, ailment, or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.